Organic Synthesis
NamedReactions
Up One Mechanism: AcetoAceticEsterCondensation |
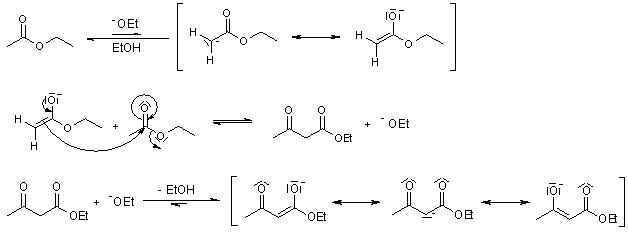 The AcetoAcetic/Claisen Condensation between esters containing α-hydrogens, promoted by a base such as sodium ethoxide, affords β-ketoesters. The driving force is the formation of the stabilized anion of the β-keto ester. If two different esters are used, an essentially statistical mixture of all four products is generally obtained, and the preparation does not have high synthetic utility. |