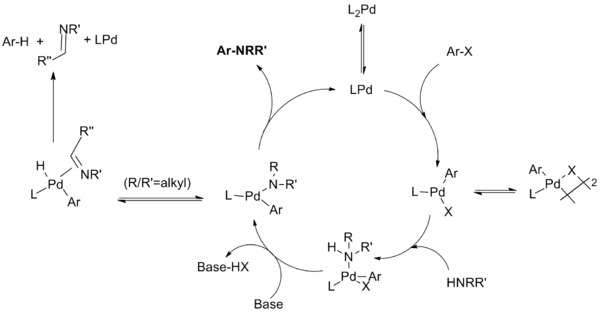
The reaction mechanism for this reaction has been demonstrated to proceed through steps similar to those known for palladium catalyzed C-C coupling reactions. Steps include oxidative addition of the aryl halide to a Pd(0) species, addition of the amine to the oxidative addition complex, deprotonation followed by reductive elimination. An unproductive side reaction can compete with reductive elimination wherein the amide undergoes beta hydride elimination to yield the hydrodehalogenated arene and an imine product.
Throughout the development of the reaction the group sought to identify reaction intermediates through fundamental mechanistic studies. These studies have revealed a divergent reaction pathways depending on whether monodentate or chelating phosphine ligands are employed in the reaction, and a number of nuanced influences have been revealed (especially concerning the dialkylbiaryl phosphine ligands developed by Buchwald).
The catalytic cycle proceeds as above image.
For monodentate ligand systems the monophosphine palladium (0) species is believed to form the palladium (II) species which is in equilibrium with the µ-halogen dimer. The stability of this dimer decreases in the order of X = I > Br > Cl, and is thought to be responsible for the slow reaction of aryl iodides with the first-generation catalyst system. Amine ligation followed by deprotonation by base produces the palladium amide. (Chelating systems have been shown to undergo these two steps in reverse order, with base complexation preceding amide formation.) This key intermediate reductively eliminates to produce the product and regenerate the catalyst. However, a side reaction can occur wherein ß-hydride elimination followed by reductive elimination produces the hydrodehalogenated arene and the corresponding imine. Not shown are additional equilibria wherein various intermediates coordinate to additional phosphine ligands at various stages in the catalytic cycle.
For chelating ligands, the monophosphine palladium species is not formed; oxidative addition, amide formation and reductive elimination occur from L2Pd complexes. The Hartwig group found that "reductive elimination can occur from either a four-coordinate bisphosphine or three-coordinate monophosphine arylpalladium amido complex. Eliminations from the three-coordinate compounds are faster. Second, β-hydrogen elimination occurs from a three-coordinate intermediate. Therefore, β-hydrogen elimination occurs slowly from arylpalladium complexes containing chelating phosphines while reductive elimination can still occur from these four-coordinate species."
|
