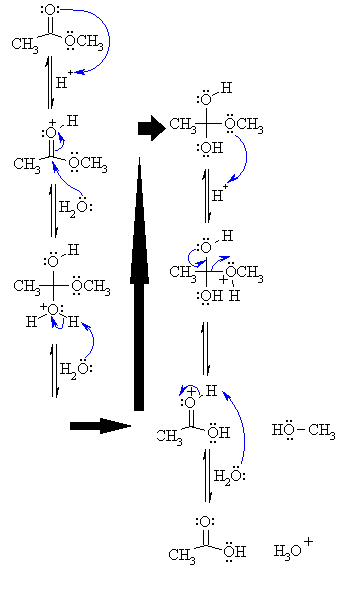
1) An acid/base reaction. Since we only have a weak nucleophile and a poor electrophile we need to activate the ester. Protonation of the ester carbonyl makes it more electrophilic.
2) The water O functions as the nucleophile attacking the electrophilic C in the C=O, with the electrons moving towards the oxonium ion, creating the tetrahedral intermediate.
3) An acid/base reaction. Deprotonate the oxygen that came from the water molecule to neutralise the charge.
4) An acid/base reaction. Need to make the -OCH3 leave, but need to convert it into a good leaving group first by protonation.
5) Use the electrons of an adjacent oxygen to help "push out" the leaving group, a neutral methanol molecule.
6) An acid/base reaction. Deprotonation of the oxonium ion reveals the carbonyl C=O in the carboxylic acid product and regenerates the acid catalyst.
|
