Organic Synthesis
Formation
| Mechanism:
|
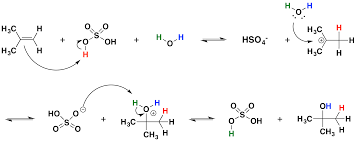 This reaction starts with a nucleophilic attack by pi bond on the hydrogen of a sulfuric acid hydrogen producing a carbocation at the more substituted carbon (due to greater stability of carbocation there). This is the rate determining step. The carbocation is then attacked by the unshared pair of electron of a water molecule, with the subsequent loss of a hydrogen ion from the attached water molecule, leaving a hydroxyl group. |
|
Return |