Organic Synthesis
Formation
| Mechanism:
|
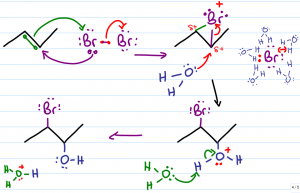 This shows the mechanism of a hydrohalogenation of an alkene that involves pi bond nucleophilic attack of a hydrogen ion, resulting in a broken double bond and a carbocation. The halide ion will add to carbocation (on the more substituted carbon) following Markovnikov's rule, due to the stability of the carbocation at that position. The product is a haloalkane, also known as an alkyl halide. |
|
Return |