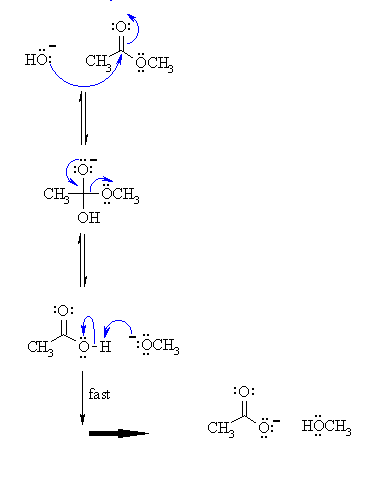
1) The hydroxide nucleophiles attacks at the electrophilic C of the ester C=O, breaking the pi bond and creating the tetrahedral intermediate.
2) The intermediate collapses, reforming the C=O results in the loss of the leaving group the alkoxide, RO-, leading to the carboxylic acid.
3) An acid / base reaction. A very rapid equilibrium where the alkoxide,RO- functions as a base deprotonating the carboxylic acid, RCO2H, (an acidic work up would allow the carboxylic acid to be obtained from the reaction).
|
